-
PDF
- Split View
-
Views
-
Cite
Cite
Stanley A Plotkin, The New Coronavirus, the Current King of China, Journal of the Pediatric Infectious Diseases Society, Volume 9, Issue 1, March 2020, Pages 1–2, https://doi.org/10.1093/jpids/piaa018
Close - Share Icon Share
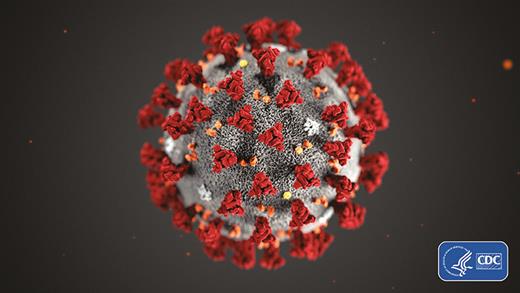
Coronaviruses are respiratory viruses whose appearance on electron microscopy looks like tiny crowns (Figure 1). The first coronaviruses discovered were the cause of pediatric and adult respiratory infections that were not particularly dangerous. One study from Vanderbilt found coronavirus in about 5% of samples from upper respiratory illness and 8% from lower respiratory illness [1]. Most clinically significant coronavirus infections have been found in children < 2 years of age, as reviewed in this journal in 2009 [2], although adults also might suffer severe infections [3, 4]. This was the picture as late as 2018: a group of respiratory viruses that could cause serious illness in the very young, but with negligible mortality [5, 6].
The ultrastructural morphology exhibited by the 2019 novel coronavirus. Photo used with permission from the Centers for Disease Control and Prevention, Alissa Eckert, Dan Higgins (available at https://phil.cdc.gov/Details.aspx?pid=23311).
Two events changed that picture: the discovery of Middle Eastern respiratory syndrome (MERS) and the explosive outbreak of severe acute respiratory syndrome (SARS) [7]. MERS causes a respiratory illness in dromedaries that can be transmitted to humans, in whom the infection can be serious and fatal [8–11]. Moreover, serologic studies show that MERS infection is common in Saudi Arabia, where contact with camels is common but also where human-to-human infection occurs [12]. There are several candidate vaccines against MERS being developed by an international organization, the Coalition for Epidemic Preparedness (CEPI; see below) [13].
As is well known, SARS causes a highly fatal infection in humans. Research on the latter virus concluded that SARS is likely a virus with a reservoir in bats [14], which can infect small mammals such as the palm civet. The latter are considered to be a delicacy to eat by some Chinese, and are sold in many markets in China. The source of SARS is thought to be from a market in Guandong. However, as the receptor for coronavirus is present in human lungs, direct infection from bats may be possible [15], all the more so because the bat coronavirus that has been isolated is genetically close to SARS [16]. Although SARS no longer circulates in humans, candidate vaccines were developed but never licensed [17–21].
Nature frequently repeats itself, and a new coronavirus, named SARS coronavirus 2 (SARS-CoV-2), has escaped from a market in the city of Wuhan in central China and is spreading from there. Although mortality seems to be lower than in the case of SARS, many of the infections cause lower respiratory tract disease requiring hospitalization. Although yet to be confirmed at this writing, it is likely that SARS-CoV-2 originated from infections transferred from small mammals in the Wuhan market to humans. In any case, it is now spreading by human-to-human contact. Respiratory secretions of infected individuals contain virus, and there is some evidence that virus may be excreted in stool. Obviously, much information is unpublished but is being exchanged through the World Health Organization (WHO) and websites. Sequences of SARS-CoV-2 show 82% homology with SARS and there is some cross-neutralization by SARS antiserum against SARS-CoV-2, but probably not enough to be useful in protecting human populations [14].
Information about the new virus is evolving rapidly, as laboratories throughout the world are working hard on it. Among the critical questions are:
How to grow the virus? (It does grow in some cell cultures.)
Are antibodies to the virus protective or enhancing?
What animal models can be used to study the virus?
Do antibodies to SARS or MERS protect against SARS-CoV-2?
What is the best vaccine antigen: whole virus, spike protein, or receptor-binding domain of the spike protein?
Can large amounts of a vaccine be made before next winter, assuming that SARS-CoV-2 becomes a seasonal threat?
For obvious reasons, a single-dose vaccine would be preferable to a vaccine requiring several doses. The current focus is to develop antibodies to the spike surface protein and in particular to the receptor-binding domain on that protein. The most obvious platforms to develop single-dose vaccines are attenuated viruses, DNA plasmids, and messenger RNA constructs coding for viral proteins that stimulate neutralizing antibodies. However, subunit protein vaccines are also being developed. The appearance of SARS-CoV-2 emphasizes that new agents will emerge from time to time to threaten humans and that mechanisms of rapid response should be in place beforehand. That was the reason CEPI was developed and, indeed, CEPI and the WHO together are marshaling large-scale preventive and therapeutic responses to the new virus [22–25].
Aside from the development of vaccines against SARS-CoV-2, a step the Chinese government should take is to ban the sale of small mammals in their markets to prevent the introduction of other coronaviruses or, for that matter, pathogens in general that could adapt to humans.
Note
Potential conflicts of interest. Dr. Plotkin consults for many vaccine manufacturers and biotechnology organizations. The author has submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
World Health Organization.